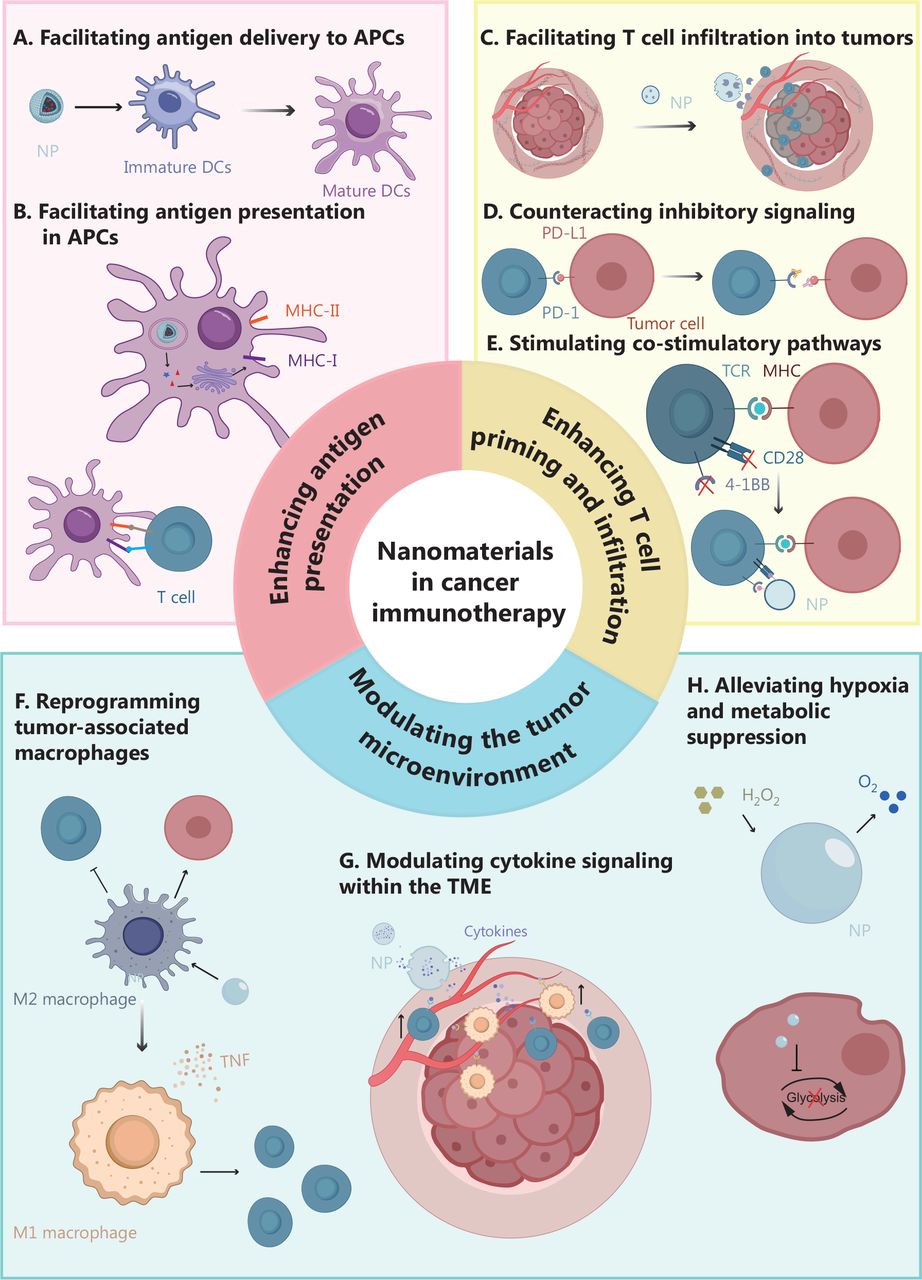
GA, UNITED STATES, May 17, 2026 /EINPresswire.com/ — Cancer immunotherapy has changed the treatment landscape, yet many tumors still evade immune attack by hiding antigens, blocking T cell entry, and building an immunosuppressive tumor microenvironment (TME). A new review brings these scattered barriers into one mechanistic framework, showing how engineered nanomaterials can be designed to intervene across the cancer-immunity cycle. Rather than acting only as drug carriers, nanoparticles (NPs) can help deliver antigens and immune stimulants, improve antigen presentation, support T cell priming and infiltration, and reshape hostile tumor niches. The review provides a practical design map for developing more precise, durable, and safer nano-immunotherapies.
Cancer immunotherapy works by mobilizing the body’s own immune system to recognize and destroy malignant cells. Immune checkpoint inhibitors (ICIs), cancer vaccines, adoptive cell therapies, and cytokine-based treatments have produced durable responses in some patients. However, clinical benefit remains uneven, partly because many tumors are “cold,” with weak antigen presentation, limited immune-cell infiltration, and strong suppressive signals. Systemic immune activation can also cause toxicity, while cell-based therapies remain expensive and difficult to scale. Based on these challenges, there is a need to carry out in-depth research into nanomaterial design principles that can improve immune activation while reducing off-target effects.
Researchers from the State Key Laboratory of Medicinal Chemical Biology, Key Laboratory of Functional Polymer Materials Ministry of Education, College of Chemistry, Frontiers Science Center for New Organic Matter, and Academy for Advanced Interdisciplinary Studies at Nankai University published this review in Cancer Biology & Medicine in April 2026. The article examines how nanomaterials can be rationally engineered to overcome delivery barriers and strengthen cancer immunotherapy by targeting key steps of the cancer-immunity cycle.
The review first explains why delivery remains a central obstacle for nanomedicine. Many NPs rely on the enhanced permeability and retention (EPR) effect, but tumor accumulation is often heterogeneous in patients. To improve precision, researchers have developed active targeting strategies using ligands, antibodies, or receptor-recognition motifs that guide nanoparticles (NPs) toward tumor cells, antigen-presenting cells (APCs), dendritic cells (DCs), macrophages, or T cells. The review then highlights intracellular design strategies, including proton sponge effects, membrane fusion, and direct translocation, which help cargos escape endosomes and reach functional sites. Across the immunity cycle, NPs can co-deliver tumor antigens and adjuvants to DCs, enhance major histocompatibility complex class I (MHC-I)-mediated cross-presentation, and promote cytotoxic T lymphocyte (CTL) activation. Other nanoplatforms are designed to block programmed death 1 (PD-1)/programmed death-ligand 1 (PD-L1) signaling, deliver messenger RNA (mRNA) encoding co-stimulatory molecules such as OX40, or carry clustered regularly interspaced short palindromic repeats (CRISPR)/CRISPR-associated protein 13a (Cas13a) systems to disrupt immune escape pathways. The review also describes approaches to reprogram tumor-associated macrophages, deliver cytokines such as interleukin-12 (IL-12), relieve hypoxia, and reduce metabolic suppression.
The authors said nanomaterials should be viewed as immune-engineering tools, not simply miniature containers. By tuning particle size, surface chemistry, degradability, targeting ligands, and release behavior, researchers can decide where an immune signal goes, when it is released, and which cell type receives it. They said the future of nano-immunotherapy will depend on matching material design with immune mechanism, so that each platform solves a defined biological bottleneck rather than adding complexity for its own sake.
These insights could guide the next generation of cancer immunotherapies toward more selective and patient-responsive designs. Nanomaterials may help turn cold tumors into inflamed tumors, expand the number of patients who benefit from immune checkpoint inhibitors (ICIs), and reduce the toxicity associated with systemic immune stimulation. At the same time, the review emphasizes that translation will require stronger evidence on tumor accumulation, intracellular trafficking, long-term safety, manufacturing consistency, and reproducibility. If these challenges are addressed, nano-immunotherapy could become an important bridge between molecular engineering and precision oncology.
References
DOI
10.20892/j.issn.2095-3941.2025.0737
Original Source URL
https://doi.org/10.20892/j.issn.2095-3941.2025.0737
Funding information
This work was supported by the National Natural Science Foundation of China (NSFC; Grant Nos. 52525310, 52373143, 22077073, and 52203172), the Tianjin Science Foundation for Distinguished Young Researchers (Grant No. 24JCJQJC00290), the Key Projects of Tianjin Science and Technology (Grant No. 24ZXZSSS00150), the Academy for Advanced Interdisciplinary Studies, Nankai University (AAIS, NKU), and the Frontiers Science Center for New Organic Matter, Nankai University (Grant No. 63181206).
Lucy Wang
BioDesign Research
email us here
Legal Disclaimer:
EIN Presswire provides this news content “as is” without warranty of any kind. We do not accept any responsibility or liability
for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this
article. If you have any complaints or copyright issues related to this article, kindly contact the author above.
![]()
Media gallery